Part Two: Corrosion is one of the most common and costly problems faced by recreational boat owners. Whether you own a trailer boat, a cruising yacht, plug into shore power or berth on a swing mooring, the marine environment constantly works against the metals on your vessel. Jessica Gatt advises how to protect against it.
The good news is that with the right knowledge and a proactive approach, most corrosion problems can be reduced or prevented entirely. In the last edition, Part One, we went through the common types of corrosion affecting boats and selecting the correct anode (seewww.marineprosystems.com/anodeguide). By understanding the mechanism of corrosion, means that the best approach to minimise or reduce the risk of that
corrosion from occurring can be undertaken. We will go through the main corrosion risks for the type of boat so you can easily navigate what is specific to you.
Aluminium boats are strong, lightweight, and popular for fishing, offshore use, and general recreation. However, aluminium can be very sensitive to electrical & galvanic corrosion. As the hull is both conductive and a higher potential material, aluminium is at risk from both galvanic corrosion and electrically induced corrosion.

Boats kept in the water generally have a higher risk of the aforementioned due to constant
submersion, so considerations for protective coatings, cathodic protection (anodes) and electrical design are absolutely crucial. Importantly, and often overlooked, is the maintenance. Most commonly aluminium boats corrode from the inside out – the effect of salt and moisture sitting on a bare hull can indeed be extremely aggressive, causing localised pitting and oxidation. The simplest way to reduce overall corrosion risk no matter where you keep your vessel, is to use a pH Neutral salt-removing boat wash and regularly
protect with an appropriate soft sealant. Maintaining the integrity of any topside paints will benefit from a similar process, with the addition of an insulating wax particularly on the paint, and the paint edges to minimise the opportunity for underfilm corrosion.
Don’t forget to similarly flush any bilges with a salt removing wash, and if kept on a trailer, open the bung and keep it as dry as possible.
For galvanic corrosion protection, this is referring to your anodes, the most important factor here is the anode type. Most commonly, outboards are fitted by the OEM with aluminium anodes (don’t worry, it’s a higher potential than the hull material – actually, it’s more active than zinc). The anode material fitted on the hull must match the type on the outboards as all systems are common. If you were to put zinc on the hull, the drive anodes will do all of the work and the zinc will passivate and fail to provide any protection. This will result in rapidly depleting outboard anodes, as they will be protecting more surface area than they are designed for.

Also importantly, regardless if your boat is kept on a trailer or in water, if your anodes are in good shape, they should be regularly removed, contact points & anode face cleaned and re-seated with Conductive Grease to ensure good connection. Any oxidation at these points can create impedance, and a failure to protect will result. We also recommend that outboards are flushed with fresh water and a salt-removing engine flush after each use to minimise the potential for any internal corrosion action. Internal anodes within the outboards are only working when the engine is in operation and full of water.
Aluminium boats kept in the water should always consider coatings on the hull, not only for protection against growth, but for corrosion protection. Minimising wetted surface area of metal material will reduce the overall surface area requiring protection from anodes. It doesn’t remove the need for cathodic protection, but does substantially reduce the anode volume required for reliable and long term protection. Antifouls should always be aluminium safe. Be very cautious using copper-based antifouls as the epoxy barrier coatings are integral in ensuring that no localised galvanic corrosion occurs between the antifoul and paint, which can be incredibly aggressive. Unless you’re substantiating the thickness and quality of the protective barrier regularly, don’t risk it.
Maintenance and integrity of the electrical system is also super important to make sure we minimise the risk of stray current and circulating currents to the hull. Keeping your electrical system in good condition, regularly inspecting for any corroded or loose cables. Keeping everything tight, free from corrosion and protected from water ingress is imperative in ensuring that the hull is not used for conduction of power, nor is there an opportunity for stray current looking for alternative paths of current – which can cause extremely aggressive hull and/or drive corrosion. If corrosion appears suddenly or worsens quickly, it is often worth having the electrical system checked for stray current.
Fibreglass hulls themselves do not corrode, but the metal components attached to them do.
Outboards fitted to fibreglass boats are usually protected by aluminium anodes, so again, any additional hull anodes that are bonded to through-hull fittings or other materials must also be aluminium anodes. Ensure that the transom assemblies are bonded to this additional anode to establish equilibrium between the two anode systems.
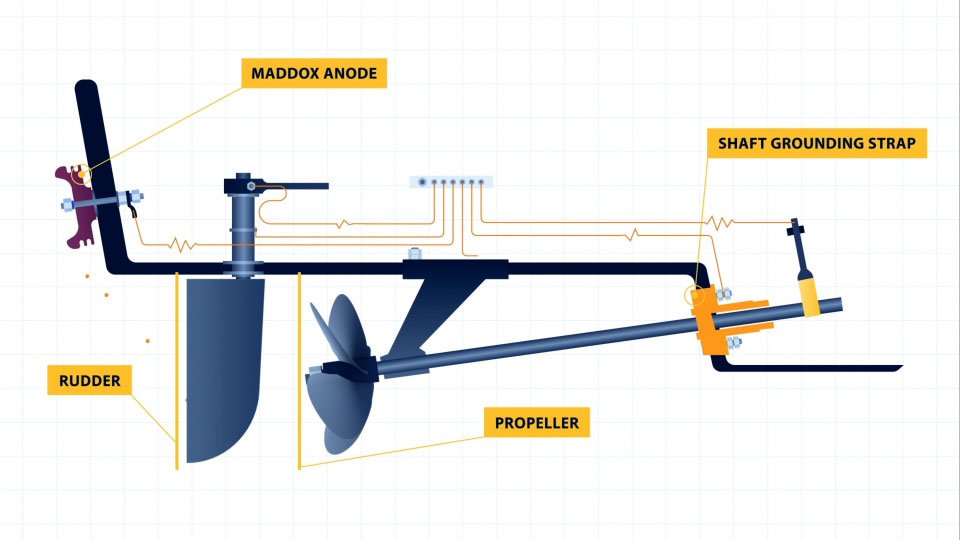
On fibreglass boats, stainless steel trim tabs can be affected by overprotection – excessive paint loss and heavy growth, all markings of excessive anode actions. Changing the anode to a more appropriate anode, the Maddox Anode, will reduce any effects of overprotection. Just make sure that the trim tabs are not bonded (isolated) so there is no interruption by the aluminium anodes.
Power boats (shaft drive and IPS) are usually fitted with hull anodes and a bonding system. A bonding system is the common earthing system that connect all underwater submerged metals to an anode for cathodic protection. It is important to note that most production sailing yachts do not have bonding systems. Also some drives, like IPS, are intentionally isolated and protected by their own protection system. So be very careful with the idea of “bonding everything” and consult a specialist before you make any changes to a bonding system.
Essentially a stray current issue has the ability to travel through the bonding system and has the capacity to affect any article that is bonded and submerged. This is why anodes are generally the first sign of an electrically induced corrosion event – they tend to wear quickly and corrosion (metallic loss) will occur regardless of the presence of anodes and regardless if the material is more active or noble.
A galvanic corrosion issue, on the other hand, is generally a slower process, and will be seen by deterioration of the most active material as it becomes the anode due to a lack of connection to a proper sacrificial anode. Think bronze turning pink on a propeller or rudder, or the alloy components on a sterndrive deteriorating – but the noble material, like stainless steel, is untouched. In this case, the connection of the metals to either the locally fitted anode or to the bonding system needs to be tested.

Usually breakdown in the bonding system, or isolation of the anodes has occurred.
A good bonding system is characterised by high-quality multistrand tinned copper cable (green/yellow is standard) at a minimum of 6mm 2 , tinned copper lugs with waterproofing around the connection and a quality fixture method to the article for protection. Ideally, star network distribution, with limited daisy chain connections to ensure reliable & serviceable connections with reduced likelihood of failure.
Exposed wires, pure copper cable, stiff and corroded cable all indicate likely potential for impedance and failure.

The benefits of bonding is providing cathodic protection to multiple parts via an electrical connection to a simple anode system. However in establishing cathodic bonding, you are also connecting all the metals to the onboard electrical system – so any onboard electrical influences can be shared to all submerged metals. But without, galvanic corrosion is a real risk, where corrosion failure of through-hull fittings, for example, can cause a sinking risk.
Maintenance of the electrical system and bonding is imperative to minimise stray current corrosion and galvanic risks. Any loose wires, localised corrosion on connections or degraded cable (stiff, poor condition of insulation sheath) will affect the bonding system performance, potentially leading to bonding failures. Similarly, the same poor condition in power cabling can indicate risk for stray currents, with the most common cause of significant DC related stray current corrosion caused by a breakdown in negative battery cabling and connections.

Signs that your anode system is not performing correctly are: excessive wear (i.e. 50% of anode mass not remaining at 12 months) – can indicate a lack of anode mass or an electrical influence. Minimal to no wear can indicate connection problems. Dependant on a number of factors, an anode should be wearing 10 – 50% by weight over 12 months. Electrical stray currents can either significantly exacerbate anode wear, or do the opposite – where there is no wear, but an active corrosion process.
Sterndrives are common on many recreational boats and are one of the most corrosion-sensitive setups. This is because they combine alloys of aluminium, stainless steel, and other metals. They are prone to coating failure, high levels of marine growth and corrosion problems. Like outboards, sterndrives are usually fitted with aluminium anodes, so if a hull anode is used – this must be bonded to the transom assemblies and be the same anode material as what is installed on the drives.
We routinely suggest at a minimum 6-monthly lift and hold to ensure the condition of the anodes and coatings, check for any degradation and clean any growth. Within the transom assembly, check the cabling for tilt/trim for any signs of damage to the cabling – this is a common source of stray current when damaged.

Shore power is a common point of blame for corrosion issues. Commonly it is thought that marinas can be “hot”, or the old boat next to you – is delivering stray current to your boat. If we take it back to understanding the mechanism of action, this helps to provide some context for minimising the risk.
In Australia, New Zealand and North America, shore power is supplied with a Common Earth – in that all pedestals share a common earth back to the main distribution. This gives the marina an Earth Potential, usually about -350mV, being the value of copper to ground. As soon as you plug in (not even turn power on), the earth on your boat becomes continuous with the marina earth, so you are a part of the common ground.
Now, before you plug in, your earth potential of your boat is the value of your earthing system, which is usually your anode potential. So if you have an aluminium boat fitted with aluminium anodes, your potential is about -1050mV (very anodic). If you’re a fibreglass boat fitted with zinc anodes, your potential is about -950mV. Simply by plugging in, you are creating a galvanic coupling – where the most active material will become the anode as it equalises with the shore earth potential. This results in high anode wear, and when the anodes are gone, it moves on to the next highest potential material in the common circuit.
So you can have a shore power influence regardless of the presence of a “stray current”.
It is this common earth that does also have the ability to share a stray current from another boat. Or share your stray current problem to the marina! This is why the Standards in these regions stipulate the requirement for a galvanic isolator when plugging into shore power. What these units do is isolate any extra low voltage DC current from flowing in or out of a vessel through the common earth, but still allow for an effective AC earth connection for electrical safety. If you plug into shore power, you need to fit an approved galvanic isolator at a minimum. Check its operation annually to make sure you’re protected.
The next step up from this is an isolation transformer – where active, earth & neutral are all physically isolated. Isolation transformers are more common in very large vessels, travelling vessels and commercial applications – they are significantly more expensive and heavy, and in smaller recreational craft are generally unnecessary.
Protective coatings are an incredibly powerful mechanism of corrosion protection – through isolation of material from seawater and oxygen and also through the reduction of available surface area that requires cathodic protection, thereby improving protection efficiencies. As corrosion generally requires oxygen and water, coatings are a simple way to reduce corrosion potential. Like anything – the choice, application methodology and maintenance of the coating is imperative for longevity and performance. We routinely recommend coatings on submerged metals and on bare materials within bilges that are not regularly maintained – it just gives you more leeway and opportunity for protection.
For aluminium vessels – bare aluminium can be subject to environmental oxidation and pitting just through salt remaining on the surface. Soft protective coatings (like waxes and appropriate lanolin-type products) can be a simple way of extending holdout against the environment and make cleaning easier. Permanent coatings obviously last longer, but like anything require maintenance. Salt, UV and chemicals on the surface of the coating can reduce its longevity – so maintaining the coating through a regular schedule of cleaning and protection with quality waxes should see significant life and performance extension.
Corrosion is an inevitable part of the marine environment, but it doesn’t have to be an expensive or unexpected problem. With the right anodes, sound electrical design, protective coatings, and a consistent maintenance routine, boat owners can significantly reduce the risk of corrosion and extend the life of their vessel and equipment. A proactive approach, combined with regular inspections and early intervention, will always be the most effective and cost-efficient form of protection on and off the water.



